European Foulbrood by Dr Chris Palgrave
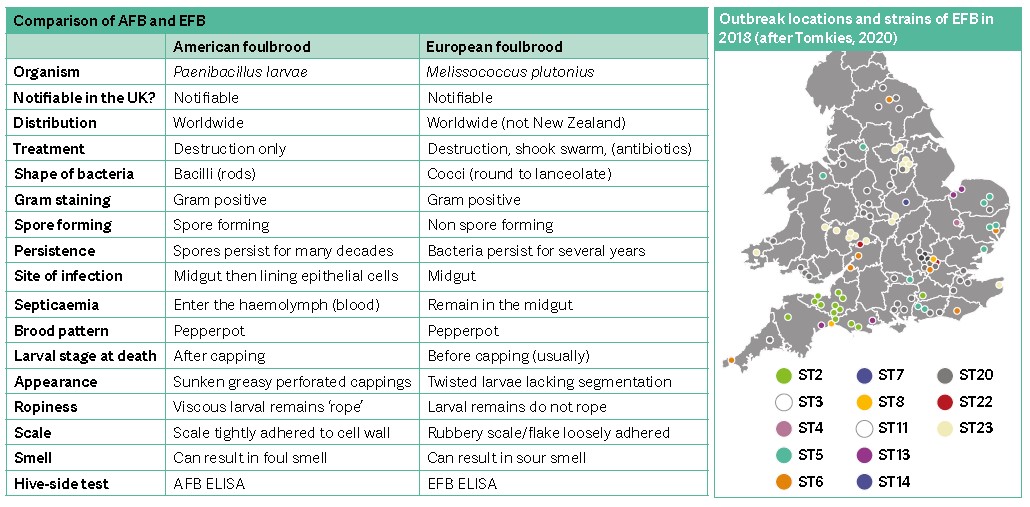
The two foulbroods, American foulbrood (AFB) and European foulbrood (EFB), are highly infectious diseases of honey bees found worldwide; they are both notifiable in the UK. Although they have likely been recognised since antiquity, their presence has been felt more keenly since the introduction of modern beekeeping practices in the mid-19th century (Tihelka, 2020). Foulbrood was once considered to be a single disease, but although the two diseases do share a number of key features, they are quite different.
In the March 2020 issue, I discussed AFB, arguably the more serious disease which has highly resistant spores that remain viable for decades. However, AFB incidence is now relatively low in the UK, in part due to rapid identification and a robust and effective control strategy.
Clinical signs
 ‘Pepperpot’ brood pattern Empty cells spotted around the brood where larvae have been removed by the worker bees. The queen may have infilled these areas with eggs. Although the pepperpot brood pattern is an important indicator of brood disease, it is not specific for EFB. It may also be associated with AFB, varroosis, chalkbrood, other brood diseases and starvation.
‘Pepperpot’ brood pattern Empty cells spotted around the brood where larvae have been removed by the worker bees. The queen may have infilled these areas with eggs. Although the pepperpot brood pattern is an important indicator of brood disease, it is not specific for EFB. It may also be associated with AFB, varroosis, chalkbrood, other brood diseases and starvation.

Twisted abnormal larvae Instead of seeing C-shaped, pearly-white larvae with distinct segmentation, infected larvae are often twisted along the cell wall, yellow or brown and lacking normal segmentations. In some cases, the distended bacteria-laden midgut can also be seen (photos from BeeBase).
 Larval remains Dead larvae eventually disintegrate (‘melt down’) and become liquid, often with an unpleasant smell. The smell is due to secondary bacteria which can overwhelm M. plutonius and confounded early attempts to identify the causative agent. The melted-down material dries into a rubbery scale or flake (as above) that sits on the lower surface of the cell indicating that something may be wrong.
Larval remains Dead larvae eventually disintegrate (‘melt down’) and become liquid, often with an unpleasant smell. The smell is due to secondary bacteria which can overwhelm M. plutonius and confounded early attempts to identify the causative agent. The melted-down material dries into a rubbery scale or flake (as above) that sits on the lower surface of the cell indicating that something may be wrong.
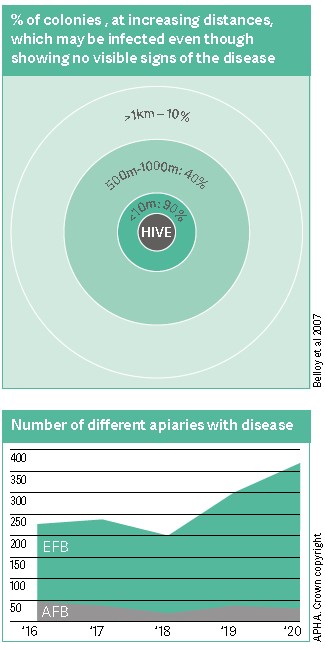 EFB is a particularly difficult disease to control because colonies may remain asymptomatic (no visible signs) for long periods of time during which the disease ‘smoulders’ unnoticed, but is spreading nonetheless. At some point, infection flares, resulting in significant clinical disease (Bailey, 1991). EFB is highly infectious and over 90% of colonies within a 10m radius of a symptomatic hive can also be infected. Alarmingly, almost 40% of colonies within a 500m and 10% within a 1km radius may be infected even though they may show no visible signs of the disease (Belloy, 2007). What is particularly concerning is that cases in England and Wales are steadily increasing year on year.
EFB is a particularly difficult disease to control because colonies may remain asymptomatic (no visible signs) for long periods of time during which the disease ‘smoulders’ unnoticed, but is spreading nonetheless. At some point, infection flares, resulting in significant clinical disease (Bailey, 1991). EFB is highly infectious and over 90% of colonies within a 10m radius of a symptomatic hive can also be infected. Alarmingly, almost 40% of colonies within a 500m and 10% within a 1km radius may be infected even though they may show no visible signs of the disease (Belloy, 2007). What is particularly concerning is that cases in England and Wales are steadily increasing year on year.
Young honey bee larvae may be exposed to Melissoccocus plutonius in contaminated broodfood, approximately 1–2 days after hatching. The bacteria multiply within the midgut, competing with the growing larvae for food (Forsgren 2010).
Well-fed larvae seem to be able to survive as long as the nurse bees can provide sufficient food for both the host and bacteria. However, surviving larvae void their faeces in preparation for pupation and contaminate the comb.
Underfed larvae will starve, typically before the cell is capped (4–5 days after hatching). The amount of brood, the food available and the number of nurse bees influence how fast the disease takes hold. Even where there is plenty of food and nurse bees, the contamination of the comb and the transfer of bacteria to increasing numbers of larvae may eventually mean that the nurse bees cannot deliver sufficient food to all the infected larvae and the disease takes hold.
By becoming familiar with what healthy larvae look like, you can recognise when things are abnormal. Even though you may detect disease during routine inspections, some specific disease-inspection visits are worthwhile. On these disease-inspection visits, work methodically through every frame, shaking off all the bees and focus on the brood.
Hive-side test
A dead larva can be tested in the field using a simple lateral flow device, from Vita Bee Health. This works in the same way as a pregnancy test (and some covid-19 tests): there is one control line (that should always appear) and one test line (which will appear only if the sample is positive).
Laboratory testing
Infected or suspect brood combs may be submitted to the National Bee Unit (NBU) for further testing.
Sequence typing
Since 2014, the NBU and Fera (Food, Environment and Rural Affairs) have compared the DNA sequence of the EFB bacteria to identify different genetic strains or ‘sequence types’ (STs) (Haynes 2013).
Approximately 35 different STs have been identified worldwide, of which about 20 are present in the UK. ST can provide valuable information about disease transmission. For example, if a particular ST is geographically distinct and suddenly appears in a new location, a movement of bees and/or equipment between the two areas is suspected. Some STs are also associated with levels of virulence or how they respond to different control measures – which can be helpful when selecting treatment options (Budge, 2014; Nakamura, 2016).
However, ST data has its limits. Some virulence factors (eg the ability to produce specific toxins) are exchanged directly between bacteria but are not part of their main chromosomal DNA (Grossar, 2020).
 Treatment options
Treatment options
EFB is a notifiable disease and must be reported to a bee inspector or the NBU. Immediately, reduce the hive entrance to prevent robbing (as the colony may be weak) and do not remove any bees or equipment from the apiary. If EFB is confirmed, a standstill order on the apiary is imposed and the bee inspector will discuss treatment options (NBU 2017 a,b).
1 Shook swarm
If it is spring/summer, the colony is strong and/or has less than 50% of the larvae infected, the inspector will probably recommend a shook swarm. A clean hive with fresh foundation is set up and all the adult bees are shaken into it – the presence of only adult bees and the queen resembles a swarm, hence the name. In doing this, the vast majority of bacteria will be removed. The colony will draw out new foundation and re-establish a brood nest and stores. The old brood combs are destroyed, and the hive components thoroughly cleaned and disinfected before reuse.
2 Destruction of colony
It is late in the season, if the colony is weak and/or has more than 50% affected larvae, the bee inspector may recommend destruction of the colony because the bees will not have sufficient time to draw out the foundation, re-establish a brood nest and gather sufficient stores for winter. Even earlier in the year, a weak colony may not have the numbers or capacity to re-establish the brood nest, so destruction may be the best option. Again, brood combs should be destroyed and hive parts disinfected.
3 Antimicrobial therapy
In certain rare circumstances, an antibiotic (oxytetracycline) may be used to help control the disease. This could be during a period of prolonged bad weather or late in the season. A shook swarm would then be performed at the earliest available opportunity (which may be the following spring).
The choice of treatment may be influenced by the ST present. Of course, destruction of the colony provides the most effective method of control, but shook swarming is moderately effective; antibiotic treatment is the least effective (Budge, 2010).
Given how infectious the EFB organism is and how easily it can be spread around the apiary, all other colonies should be carefully examined. Neighbouring apiaries will also be inspected – one of the many reasons why it is so important to register on BeeBase.
Bee Disease Insurance Ltd (BDI) and the NBU are currently running a joint initiative to perform a whole apiary shook swarm to reduce re-occurrence of EFB (even if there is no visible evidence of infection in some of the colonies).
Contributing factors
Many factors may contribute to trigger an asymptomatic colony to become symptomatic.
The ratio of nurse bees to larvae (and the impact on their ability to feed the larvae) is likely to be of most importance. Outbreaks are often seen in the spring and are probably related to the rapid expansion of the brood nest when there are relatively few overwintered nurse bees. Stores may also be running short and the colony is more vulnerable to sudden cold snaps. The overwintered nurse bees are also susceptible to co-infection, including varroa mites (and the viruses they transmit), sacbrood virus (which can affect the hypopharyngeal brood-food glands) and nosema (see BeeCraft May 2020). Spring manipulations (eg splitting colonies, spreading the brood, stimulatory feeding) may result in an imbalance of nurse bees to larvae or affect their ability to feed them and maintain the temperature of the brood nest. Split colonies may abruptly lose many foragers and with it an incoming food supply.
There are many non-specific stressors which can disrupt the delicate balance of a colony. Although some factors, like weather, are beyond our control, we can make a difference by considering the frequency, manner and length of our inspections and manipulations. However keen we are to see our bees (and we have all been super-enthusiastic new beekeepers), I would suggest that it is best to follow three cardinal rules:
1 Only go into a hive if you have a good reason to
2 Have a clear plan about what you may do
3 Avoid unnecessary manipulations.
ROB Manley, the influential 20th century British bee farmer and author, with a twinkle in his eye, would refer hobbyists to TW Cowan’s The British Beekeeper’s Guide Book which has “all the fads and unnecessary trimmings that so delight the hearts of enthusiasts who want to ‘keep bees’… The poor little creatures to be commenced upon in March and ceaselessly tormented until winter brings them respite.”
Dr Leslie (Bill) Bailey, renowned bee pathologist of comb-change fame, thought that “the most important means of defence insect populations have … are the ones most easily upset by beekeeping practices” (1977). And, in April this year, Ann Chilcott, Scottish Expert Beemaster, quoted a retired Government Lead Bee Inspector for Scotland: “Beekeepers kill more bees than disease in Scotland.”
We can do so many things with our colonies to maximise productivity, but I would urge hobbyists to ask themselves if a particular manipulation is really necessary? Most of us are not under the commercial pressures of bee farmers, so we have an opportunity to look afresh at our beekeeping priorities.
Finally, locality is all-important. All beekeeping is local and in some parts of the country even adjacent valleys can have wildly different ecosystems. Make use of your local associations, your local mentors, experienced colleagues and experts. I, for one, still have much to learn.
Prevention
EFB presents a particular challenge as it is complex and still not fully understood. As with any infectious disease, the usual recommendations about good sanitary practice to prevent spread should be followed. Careful apiary design to prevent drifting and effective colony management to minimise robbing will also help to reduce transmission of EFB within and beyond the apiary.
NBU's 10 rules for control
See Foulbrood disease of honey bees and other common brood disorders on BeeBase.
1 Be familiar with the signs and causes of foulbrood.
2 Carry out specific brood disease inspections (at least twice a year).
3 Never transfer combs to other colonies without checking them.
4 Never bring colonies, combs or equipment into the apiary without checking them.
5 Never buy old combs. Disinfect second-hand equipment.
6 Never leave combs exposed to robbing bees. Never feed honey from another source to your bees.
7 Seal the hives of any colonies that die to prevent robbing before examination.
8 Inspect any colony that is failing to thrive for brood disease.
9 Beware of swarms. Hive on foundation in an isolation apiary.
10 Regularly and systematically replace old brood combs.
Dr Chris Palgrave BSc (Hons) BVM&S PhD FHEA FRCPath MRCVS
Chris Palgrave is a veterinary surgeon and specialist in veterinary pathology. He is a beekeeper and member of the British Bee Veterinary Association. He has worked in private practice, academia and industry, and teaches in veterinary schools and gives talks to beekeeping associations, schools and community groups. He runs Three Hares Honey from his home in the Exe Valley in Devon.
REFERENCES
Bailey, L (1977). Good Beekeeping and Bee Health. Lecture to the Central Association of Bee-keepers, 1 October
Bailey, L (1983). Melissococcus pluton, the cause of European Foulbrood of honey bees (Apis spp.) J Applied Bacteriology 55:65-69
Belloy, L et al (2007). Spatial distribution of Melissococcus plutonius in adult honey bees from apiaries and colonies with and without symptoms of European foulbrood. Apidologie. 38:136-140
Budge, GE et al (2010). The occurrence of Melissococcus plutonius in healthy colonies of Apis mellifera and the efficacy of European foulbrood control measures. J. Invertebr Pathol, 105:164-170
Chilcott, A (2021). Why we need beekeeping education. The Beelistener, 30 April 2021. www.beelistener.co.uk
Forsgren, E et al (2005). Distribution of Melissococcus plutonius in honeybee colonies with and without symptoms of European foulbrood. Microb Ecol. 50:369-374
Grossar, D et al (2020). Putative determinants of virulence in Melissococcus plutonius, the bacterial agent causing European foulbrood in honey bees. Virulence. 11(1):554-567
Haynes, E et al (2013). A typing scheme for the honeybee pathogen Melissococcus plutonius allows detection of disease transmission events and a study of the distribution of variants. Environmental Microbiology Reports, 5:525-529
Manley, ROB (1985). Honey farming, 2nd edition. Northern Bee Books
Milbraith, M (2021). Honey Bee Bacterial Diseases. In: Kane, TR, and Faux, CM (Eds) Honey Bee Medicine. Wiley Blackwell
Nakamura, K, et al (2016). Virulence differences among Melissococcus plutonius strains with different genetic backgrounds in Apis mellifera larvae under an improved experimental condition. Sci Rep 6: 33329
National Bee Unit (2017). Foulbrood Disease of Honey Bees and other common brood disorders. APHA
National Bee Unit (2017). Statutory Procedures for controlling honey bee pests and diseases. APHA
Tihelka, E and Coppi, S (2020). A brief history of foulbrood: from superstitions to veterinary medicine. The Beekeepers Quarterly, September 2020
Tomkies, V (2020). Improving our understanding of European foulbrood (EFB) transmission. BeeFarmer, February 2020
Vidal-Naquet, N (2015). Honeybee Veterinary Medicine: Apis mellifera L. 5m Publishing
August 2021
Other articles this month included:
Are beekeepers the enemy? Prof Dave Goulson talks to Sarah Wyndham Lewis
Varroa and IPM: How to apply the principles, by Norman Carreck NDB
Pondlife: Planting with water and bees in mind, by David Robinson
Beekeeping with a KISS, by Ted Scott
Very easy honey ice cream recipe with Varsha Patel




